Imagerie fœtale
Thérapeutique
Publié le 04 juin 2019Lecture 7 min
MRI of the fetal corpus callosum with tractography: a step beyond ultrasound
Houman Mahallati1, Neil O’Gorman1, Laurent J Salomon1, David Grevent2, Anne-Elodie Millischer2
The corpus callosum develops from 7 weeks’ gestation and is completely formed by 20 weeks. It develops in four sections, starting with the anterior genu, then the body and splenium posteriorly, and finally the rostrum, which extends infero-posteriorly from the genu.
Introduction
Improved ultrasound technology now enables direct visualization of the corpus callosum in the prenatal period, especially in centers where the routine second trimester ultrasound is performed at the 20-22 weeks gestational age window. This is most easily accomplished through sagittal and coronal views through the anterior fontanel(1). Often, even if the fetus is not in an ideal position, gentle manipulation of the fetus will present an acoustic window through which high quality 2D imaging is possible. In addition, when direct visualization is not possible, modern ultrasound systems with 3D facility offer the ability to acquire 3D volumes, and subsequent multi-planar reformatting can yield diagnostic images of the corpus callosum.
Developmental abnormalities of the corpus callosum can occur in different forms such as hypoplasia, hyperplasia and agenesis. Agenesis of the corpus callosum (ACC) is typically classified as complete or partial. The prevalence of corpus callosum abnormalities varies between different studies ranging from 0,3 to 0,7% of the general population(2) and 2 to 3% in the developmentally disabled population(3). ACC has also been reported to occur with other cerebral and/or extracerebral malformations. The etiology is variable in origin with studies quoting genetic factors, alcohol consumption, maternal PKU, infections and ischemia as potential factors(4-6).
When malformations or anomalies of the corpus callosum are identified on prenatal ultrasound imaging, fetal MR imaging can aid in diagnosis and management by confirming the ultrasound findings and also evaluating for the presence of other CNS or fetal abnormalities. As with many other fetal abnormalities, prognosis will depend on the presence or absence of other fetal abnormalities, and whether or not the anomaly of the corpus callosum is associated with chromosomal or genetic abnormalities. The combination of the ultrasound, MRI, and clinical findings provides the foundation upon which counseling can be offered to patients. Often this is achieved through multidisciplinary teams including pediatric neurologists and geneticists.
Agenesis of the corpus callosum
Ultrasound diagnosis
It is difficult to ACC prior to 18 weeks’ gestation. Ventriculomegaly is often the first indirect sign that ultimately leads to this diagnosis with ultrasound. More precisely, it is a particular parallel colpocephalic configuration of occipital horns. Hence, imaging in the axial plane is important as it shows the telltale and important appearance of the ventricles(7). Final diagnosis requires direct visualization in the sagittal and coronal planes. The other indirect signs include absence of the cavum septum pellucidum, upward displacement of the third ventricle, and other midline lesions or cysts. Partial ACC affects the most posterior portion more frequently, and often has a lesser degree of anatomical distortion. The diagnosis is difficult, as it requires demonstration of a foreshortened corpus callosum with a midsagittal view. Volpe et al. found that in partial ACC, the CSP was present in 85% of cases(4), thus making the prenatal diagnosis more difficult. Doppler is very useful as the peri-callosal artery is absent in complete ACC and shortened in partial ACC(5).
MRI diagnosis
Multiplanar fetal MRI, with SSFSE T2-weighted sequences, is used to screen for additional abnormalities such as cortical malformations, parenchymal lesions, brainstem and cerebellar abnormalities. Lesions such as interhemispheric lipomata and interhemispheric cysts can be seen in association with ACC. Even if MRI can offer improved detail of the morphology of the fetal brain, classical sequences remain inadequate to fully assess the structure of the corpus callosum.
Advanced imaging of the corpus callosum
Recently published guidelines on fetal MRI focus on widely available, routine sequences that permit anatomic imaging of the fetus. However, advances in MR technology now permit the use of a number of advanced functional techniques in fetal MR imaging. One such technique is diffusion tensor imaging (DTI). While a detailed explanation of the technique of the principles of DTI is beyond the scope of this short monograph, this imaging technique takes advantage of the diffusion of water molecules and permits reconstruction of images that illustrate the anisotropic diffusion of water molecules along fibers or bundles, such as can be seen in white matter tracts. Given that DTI is essentially imaging “movement” of water molecules, fetal movements pose a significant technical challenge. In order to mitigate against fetal movements, DTI sequences have been tailored to acquire the imaging data in as short a time as possible(6).
When the brain can be imaged during a phase fetal quiescence in the absence of significant fetal head movements, DTI data sets can be obtained which permit the display of white matter bundles using post processing software.
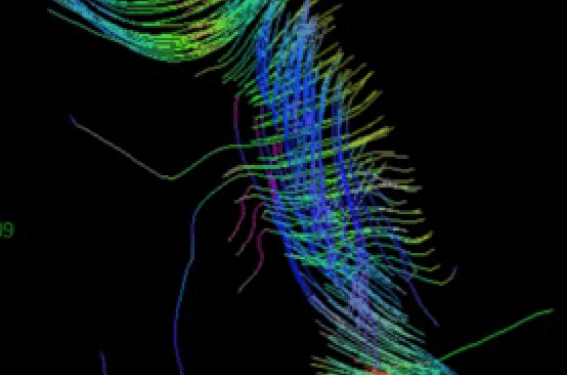
We have started to perform DTI images of the corpus callosum when feasible. While our experience to date is limited, we succeeded in a number of cases and could demonstrate, in addition to anatomic imaging, DTI data sets that show the decussating fibers across the fetal corpus callosum. In addition, given that cases with usable DTI are inherently cases with little or no fetal motion during the image acquisition, we are able to overlay the color maps of callosal fibers on conventional 2D imaging (figures 1 and 2).
Figure 1 a : Sagittal ultrasound showing a completely formed corpus callosum that measures less than the 1% for gestational age. b: Sagittal T2 weighted MR image in same patient.
Figure 2 : DTI tractography images demonstrating crossing fibers across the entire length of the corpus callosum and a fusion image showing corresponding morphology on a 2D sagittal image.
The appearance on our fetal DTI maps is very similar to those seen in post-natal and adult normal DTI maps. This impression is supported by a recent study that compared prenatal and postnatal tractography in 12 patients(7).
Clinical management
When ACC is suspected, even in the context of an otherwise normal ultrasound, karyotyping with an array CGH should be performed, as both chromosomal and genetic abnormalities are also common. The clinical relevance of callosal agenesis can pose considerable difficulties for both the fetal medicine specialist and for the patient. This is due to the difficulty in counseling patients, as the overall prognosis remains controversial. The neurodevelopmental outcome is extremely variable, even in children with similar neuroanatomic abnormalities, and in those with complete or partial ACC, there is a significant overlapping in neuropsychological performance.
A recent meta-analysis highlighted the importance of classifying the abnormality into partial or complete, as this can impact the approach to counseling. They reported that partial ACC has higher incidences of chromosomal abnormalities, additional abnormalities found on MRI and additional abnormalities found after birth(8). The overall outcome for partial ACC appears to be guarded in comparison to complete ACC (table 1).
Table 1 : Clinical outcomes associated with isolated complete and partial agenesis of the corpus callosum. Taken from D’Antonio et al.(12).
Short corpus callosum In 1993, Malinger and Zakut(8) published normal developmental growth curves for both the length and thickness of the corpus callosum using transvaginal ultrasound. There have been subsequent growth charts published since; however, they were constructed using small numbers with heterogeneous statistical approaches. In order to obtain an accurate measurement, the corpus callosum should be visualized in a mid sagittal section as an anechoic structure delineated by two echogenic lines: the superior border consisted of the sulcus of the corpus callosum and the cingulate gyrus, and inferiorly by the cavum septi pellucidi, the cavum vergae, and the lateral ventricles. The length should be measured from the most anterior aspect of the genu to the most posterior aspect of the splenium, by using a straight rostrocaudal line (figure 3).
Figure 3 : a: Sagittal image showing the corpus callosum at 27 weeks gestation. Calipers show measurement (between the 10 th and 50 th centiles for this gestational age) and b: Color Doppler image showing the peri-callosal artery.
Routine measurement of the corpus callosum is not universal and accurate measurement is highly operator dependent. In our experience, with increased imaging of the corpus callosum at the time of prenatal ultrasound, it is not uncommon to find a morphologically normal, but short, corpus callosum.
When isolated, a normal, but short corpus callosum can also present a counseling challenge(9, 10). This is in part due to the heterogeneity of the studies that report outcomes in the setting of prenatally diagnosed corpus callosum anomalies. The prognosis is often poor, as it appears to result from a late destruction of the corpus callosum within a more general destructive process. Some meta-analyses suggest that developmental delays can be found in approximately 25% of patients with isolated abnormalities of the corpus callosum, but to our knowledge, there are no studies that have specifically reviewed the outcomes in morphologically normal, but short corpus callosum. A number of metabolic and non-metabolic inherited diseases have been implicated in the etiology of a hypoplastic corpus callosum (table 2).
Table 2 : Metabolic and non-metabolic inherited diseases associated with hypoplasia of the corpus callosum. Adapted from Paupe et al. 2002(11).
In our practice, when a short, but otherwise normal, corpus callosum is identified at fetal ultrasound we routinely offer both genetic testing and counseling as well as MR imaging to patients. This MR examination is usually performed at about 30-32 weeks gestational age. The principal goals are to confirm the normal morphology of the corpus callosum and to detect additional subtle abnormalities that may not have been visible on ultrasound, such as migration anomalies.
The past two to three decades have seen a dramatic increase in our understanding of fetal growth and the importance of biometric charts constructed with both the correct population of fetuses and the appropriate sample sizes. Perhaps the time has come for us to develop more modern charts to measure the corpus callosum. In addition, given the wide array of genetic and biochemical tests with which we can now diagnose uncommon antenatal diseases, we feel that, when possible, addition of modern imaging techniques such as MRI is appropriate if we are to provide patients with the most up-to- date counseling and information. The role of tractography in these patients remains to be seen, but at present we believe that, when possible, the addition of fetal tractography to MRI of callosal dysplasia can serve as a useful adjunct, and the images may serve as a tool to assist in counseling patients.
Attention, pour des raisons réglementaires ce site est réservé aux professionnels de santé.
pour voir la suite, inscrivez-vous gratuitement.
Si vous êtes déjà inscrit,
connectez vous :
Si vous n'êtes pas encore inscrit au site,
inscrivez-vous gratuitement :